
The transport of vitamin E has long been centered on α-tocopherol as α-tocopherol has strong binding affinity to α-tocopherol transfer protein (α-TTP). Tocotrienols, on the other hand, have not been well-studied compared to α-tocopherol as tocotrienols show lack of specific affinity towards α-TTP. It has been reported that the presence of alpha-tocopherol prevents the uptake of tocotrienol. However, an animal study (in α-TTP knock-out mice) has shown that tocotrienols were transported to vital organs even without the presence of α-TTP [1]. Furthermore, like other fat-soluble vitamins and phytonutrients (A, D, K, Tocopherol, Carotenoids), the absorption of tocotrienols via oral supplementation is poor and inconsistent. In addition, the consistency or adequacy of the secretions of bile and lipases also affect the absorption of tocotrienols. Since the absolute absorption of each of the tocotrienol isoform is poor, irregular and erratic, a relatively good absorption of tocotrienol into the bloodstream is important to exhibit good bioavailability (ie: absorption of oral administered tocotrienol) and bioefficiency (ie: accumulation of tocotrienol in vital organs). Hence, one will be getting the optimal benefits that tocotrienol can provide.
Yap and colleagues studied the pharmacokinetics and bioavailability of tocotrienols via oral supplementation versus intravenous injection (IV) (α-,γ- and δ-tocotrienols). The researchers reported that the absorption of tocotrienol is influenced by the type and amount of oil vehicles.
Additionally, the same group of researchers reported that the bioavailability of α-, γ- and δ-tocotrienol was 27.7, 9.1 and 8.5% respectively when tocotrienols were administered orally. This clearly proved that the absorption of tocotrienols is poor, erratic and incomplete when consuming normal tocotrienol oil extract. The absorption of tocotrienol is associated with the solubility of vitamin E, amount of bile secretion for emulsification process and the lipolytic action by pancreatic enzymes. Therefore, tocotrienol formulation in the context of colloidal dispersion is crucial to enhance the bioavailability. Yap and Yuen embarked on further research and developed a self-emulsifying drug delivery system (SEDS) technology-“the SupraBio™ system” to overcome the low bioavailability of tocotrienols.
In this study, 12 male volunteers were recruited where 6 male subjects received orally supplemented bio-enhanced SEDS formulated tocotrienol or SupraBio™ while the other 6 male subjects received non-SEDS formulated tocotrienols. The blood samples were collected every 2-hour interval over 24-hour after dosing and the analysis of plasma levels of α-, γ- and δ-tocotrienol were conducted.When comparing to non-SEDS tocotrienols under fasted condition, SEDS-formulated tocotrienols/SupraBio™ showed
- Significant higher maximum plasma concentration (Cmax) of α-, γ- and δ-tocotrienol, up to average of 250%,
- Shorter time to reach peak plasma levels (Tmax);
- Increased bioavailability of tocotrienols by 2-3 fold
The mean plasma concentration of α-, γ- and δ-tocotrienol following the oral supplementation of SEDS-formulated 200 mg mixed tocotrienol / SupraBio™ and a non-SEDS formulated are shown in the graphs below:
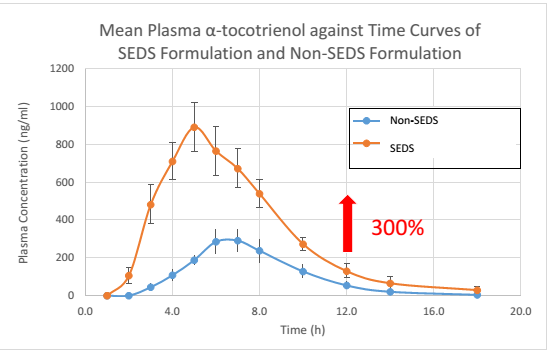
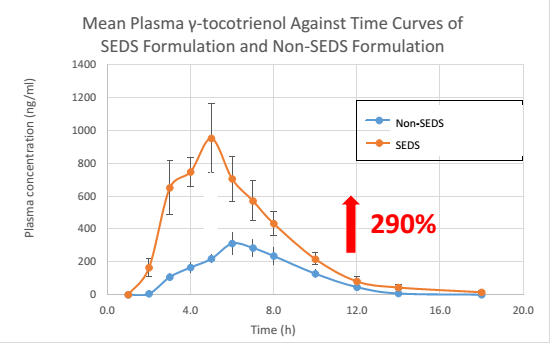

Taken together, the unique attributes of SEDS / SupraBio™ when comparing to non-SEDS are as follows:
- Absorption of tocotrienol is independent of dietary fat intake;
- Promotes absorption of tocotrienols via lymphatic system;
- Ensures a rapid transport of tocotrienols into the blood;
- Consistent & rapid absorption;
- Achieving therapeutic plasma blood level at a lower dosage due to an average of 250%increase in absorption of each tocotrienol individual [3]
Interestingly, the first ever and only actual human tissues distribution study was conducted by a group of researchers from Ohio State University, Wexner Medical Center. This NIH (National Institute of Health)-funded study has demonstrated that the oral supplemented tocotrienols were well-absorbed and transported efficiently to vital organs and tissues such as brain, liver, heart, skin and adipose tissues [4].
References
- Khanna et.al (2005). Delivery of orally supplemented α-tocotrienol to vital organs of rats and tocopherol-transport protein deficient mice. Free Radical Biology & Medicine 39; 1310-1319.doi:10.1016/j.
- Yap SP, et al. (2001). Pharmacokinetics and bioavailability of α-, γ- and δ-tocotrienols under different food status. Journal of Pharmacy and Pharmacology, 53, 67-71.
- Yap SP, et al. (2003). Influence of route of administration on the absorption and disposition of alpha-, gamma-, and delta-tocotrienols in rats. Journal of Pharmacy and Pharmacology, 55, 53-58.
- Patel V, et al. (2012). Oral Tocotrienols are transported to Human Tissues and Delay the Progression of the Model for End-Stage Liver Disease Score in Patients. The Journal of Nutrition, 142(3), 513-9.