
Fatty liver is a term of situation that describes the buildup of fat in the liver. It is normal to have some fat in the liver, but once the fat is more than 5-10% of the liver weight, it is considered as fatty liver. Fatty liver is usually asymptomatic. However, if precaution and necessary actions such as healthy diets and exercise are not practiced, the fatty liver will progress to nonalcoholic steatohepatitis (NASH) and can further progress to permanent liver damage in the form of cirrhosis or cancer. Hence, fatty liver is also known as the precursor of the metabolic syndrome.
The American Liver Foundation has revealed the latest statistic on NAFLD (Non-Alcoholic Fatty Liver Disease) and reported that about 25% of the people in the U.S. are living with NAFLD, meaning 1 in every 4 Americans is diagnosed with this chronic disease. NAFLD is one of the most common onset of chronic liver diseases and also the third most common indication for liver transplantation in the U.S.
It is strongly postulated that the remission of fatty liver could be attributed to the potent antioxidative effect of tocotrienol. Alpha-tocotrienol is 40 to 60 times more potent than alpha-tocopherol as an antioxidant. As fatty liver is an early sign of liver damage, the protective effects of tocotrienol against oxidative stress associated liver damage could be the attributing factor for NAFLD and liver cirrhosis mitigation. Several clinical studies on bio-enhanced palm tocotrienol complex have shown positive results in attenuating fatty liver, reducing liver stiffness, and maintaining healthy liver functions.
Clinical Trial: Bio-enhanced Palm Tocotrienol Complex and Non-Alcoholic Fatty Liver Disease (NAFLD)
Bio-enhanced palm tocotrienol complex has shown to cause remission of fatty liver. This interventional, double-blind placebo-controlled trial recruited 64 volunteers with ultrasound-proven NAFLD. The subjects were randomized to receive either daily supplementation of 400mg palm tocotrienol complex or placebo for 1 year. Liver function tests were normal or slightly elevated for all subjects. Alcohol intake, assessed through interview, was less than 20g/week. High sensitivity B-mode ultrasound examination was performed at baseline and after 1 year by the same experienced radiologist using the same instrument.
This double-blind, placebo-controlled clinical study demonstrates the efficacy of tocotrienol in managing NAFLD was published in 2013. Results reveal that 20 patients (66.7%) out of 30 subjects in the supplemented group show significant improvement in their fatty liver condition. Of these, 50 percent of the subjects had complete remission of their NAFLD. The results are statistically significant (P < 0.05) compared to the control group [1].
Clinical Trial: Tocotrienols Decrease Liver Stiffness among NAFLD Patients
Mitochondrial reactive oxygen radicals cause lipid peroxidation and production of apoptotic or necrotic factors in the liver. As a result, collagen formation in the extracellular matrix will be increased and may progress to liver fibrosis or liver cirrhosis. Liver Stiffness Measurement (LSM) using transient elastography determines the liver fibrosis stage in NAFLD patients.
In this clinical study, 67 patients with ultrasound-proven NAFLD were enrolled and were assigned to either daily supplementation of 100 mg of mixed tocotrienols and lifestyle modification advice group (LMAG) for 3 months or in LMAG only. After 3 months of supplementation, 57% of all patients (n=38) showed decrease in their liver stiffness measurement, but 43% (n=29) did not improve. 79% of those who improved were from the Treatment group and 21% from LMAG alone. Out of the 29 patients who did not improve, 23 patients or 79% are from LMAG alone and only 6 patients or 21% from Treatment group.
This suggested that tocotrienols, together with positive lifestyle intervention is about 4 times more effective compared to positive lifestyle modification alone in the context of liver stiffness amelioration [2].
Clinical Trial: Bio-enhanced Palm Tocotrienol Complex Improves Poor Liver Functions in Prospective Liver Transplantation Patients
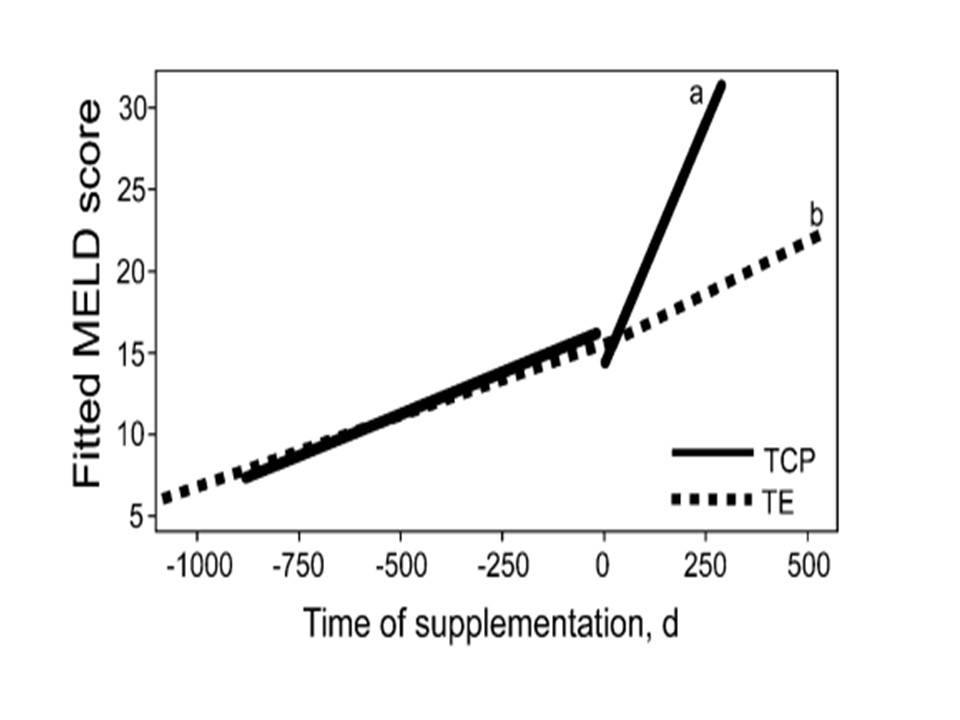
This is the very first clinical trial with proven bioavailability and accumulation of tocotrienols in vital organs following oral consumption of bio-enhanced palm tocotrienol complex. In this study, the end-stage liver disease patients were randomized to receive either 400mg of tocotrienol complex or 400mg of tocopherols. The diagnosis of the severity of liver cirrhosis utilized MELD (Model for End-Stage Liver Disease) scoring system where a poor liver condition is indicated by higher MELD score and hence the greater need for a liver transplantation and vice versa.
After 12 weeks of supplementation, 50% of the subjects supplemented with tocotrienol complex demonstrated improvement in MELD score whereas 20% of the patients with tocopherol supplementation showed reduction in MELD score. This study suggested that tocotrienol complex reveals promising results in improving poor liver functions [3].
References
- Magosso E, et al. (2013, Dec 27). Tocotrienols for normalisation of hepatic echogenic response in nonalcoholic fatty liver: a randomised placebo-controlled clinical trial Nutr J., 12(1), 166.
- Arguillas M, et al. (June 7th, 2013). The effect of vitami E (mixed tocotrienol) on the liver stiffness measurement measured by transient elastography (fibroScan) among NAFLD patients. APAS Liver Week. Singapore.
- Patel V, et al. (2012). Oral Tocotrienols are Transported to Human Tissues and Delay the Progression of the Model for End-Stage Liver Disease Score in Patients. The Journal of Nutrition, 142(3), 513-9.